#Accu chek 360 software free download trial#
The primary objective of this randomized controlled trial (RCT) is to determine the impact of bant2, an evidence-based, patient-centered, behavioral mobile app intervention, on the self-management of T2DM. We hypothesized that the effective self-management of non-insulin treated T2DM requires a behavioral intervention that empowers patients with the ability to self-monitor, understand the impact of lifestyle behaviors on glycemic control, and adjust their self-care based on contextualized SMBG data. While self-monitoring of blood glucose (SMBG) has proven to be effective for those treated with insulin, it has been shown to be less beneficial for those only treated with oral medications or lifestyle modification. Patients with type 2 diabetes mellitus (T2DM) struggle with the management of their condition due to difficulty relating lifestyle behaviors with glycemic control. Improvements in the control group may reduce effect sizes and limit statistical significance therefore, alternatives to the traditional randomized controlled trials may be considered. There are several aspects to consider in medical device studies when using interventions that rely on changes in behavior to achieve an effective implementation and significant study results.
Adaptive study designs might reduce the necessity of a separate pilot study and combine the exploratory and confirmatory stages of an investigation in one single study. Furthermore, retrospective data collection could be a feasible option. Another option is to include a third study arm to control for enhanced standard of care and study effects. In order to reduce improvements in the control group, it is essential to identify the specific reasons and adjust study procedures accordingly. Improvements in the control group can also be caused with increased attention paid to the subjects. A limited effect size due to improvements regarding clinical endpoints in the control group is often caused by the Hawthorne effect. However, randomization and blinding are not always possible.

To successfully engage patients, priority should be given to health care professionals being engaged, operational support in technical issues being available, and adherence being assessed in detail.Īnother difficulty is to avoid contamination of the control group with the intervention therefore, strict allocation concealment should be maintained.

Lessons from the STeP and ProValue studies demonstrated the difficulty in medical device studies that rely on behavioral changes in intervention group patients.
This article aims to provide challenges in interpreting diabetes management intervention studies and suggests approaches for optimizing study implementation and for avoiding pitfalls based on current experiences. Such studies thus have to be well designed in order to allocate all observed effects to the defined intervention and to exclude effects of other confounders as well as possible. However, chronic disease management is intricate with complex multifactorial behavior patterns.
#Accu chek 360 software free download how to#
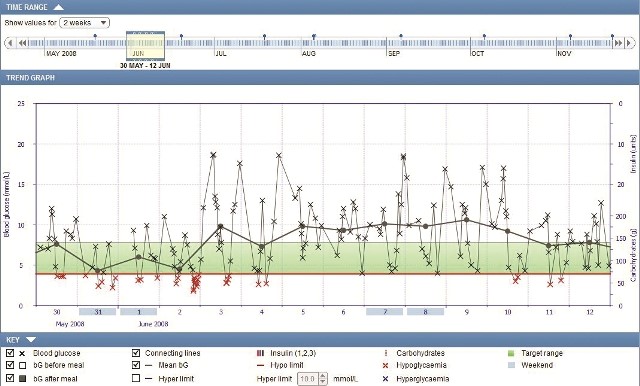
How to use the Accu-Chek 360° diabetes management systemįor more information on how to use the Accu-Chek 360° diabetes management system software, click on the Help button in the top right hand corner of the software interface, or contact (insert affiliate contact information).Several clinical studies investigated improvements of patient outcomes due to diabetes management interventions.
